Question
I- Answer as required.
(A) Use the given equation to complete
the following table and
predict which the
thermodynamic process could be spontaneous or nonspontaneous.(10 pts)
ΔGo
= ΔHo - TΔSo
|
ΔHo
|
ΔSo
|
- TΔSo
|
ΔGo
|
Comments
(Spontaneous Or Non Spontaneous)
|
Condition of T
|
|
-Ve
|
+Ve
|
...........
|
-Ve
|
|
N/A
|
|
+Ve
|
..........
|
+Ve
|
+Ve
|
|
N/A
|
|
-Ve
|
..........
|
+Ve
|
-Ve
|
|
.......................
|
|
-Ve
|
-Ve
|
+Ve
|
+Ve
|
|
.......................
|
|
+Ve
|
+Ve
|
.........
|
+Ve
|
|
.....................
|
|
+Ve
|
+Ve
|
.........
|
-Ve
|
|
......................
|
4 pt (B) Explain Why?
1-
Given that ΔG = ΔH - TΔS
And ΔH=
ΔE + W” And ΔS = q ÷ T, also
q= ΔE + W1
explain the meaning of ΔG.
2- Using the formula S = k ln W (Boltzmann equation)
to show how the
entropy of impure substance at 0.0 K is not zero.
6 pts (C) Explain the meaning of the
following:
1- ªGo
is zero:
2- n(reaction order) is zero:
3- ªHo
is an inadequate criterion of spontaneity:
Question II- Consider
the given exothermic reaction and the accompanying data when answering the
following questions.
2 H2(g) + 2 NO (g) ======= N2 (g) +
2 H2O (Kc = 90 at727 oC.)
2 pt a)
Write the Kc expression for this reaction.
2 pt b) What is the
value of Kp at 727 oC.?
4 pt c)
Use Le Chatelier's principle to predict the effect of
the following on the position of the above reaction equilibrium. ( compare Q and K in each
case)
1- Addition of some N2(g).
2-
Removal of some NO (g).
3-
Cooling the reaction container and it's contents.
4-
Decreasing the pressure.
4 pt d) For the above reaction, if >Go 727oC is -32.509 KJ, what
will be the value of Kp .
6 pt e) Suppose the following
mechanism is correct for the above reaction.
step
1 2 NO ==== N2O2 fast
step 2 N2O2 + H2 =====
N2O + H2O slow
step 3 N2O + H2 =====
N2 + H2O fast
Write the reaction rate (law)
equation.
2 pt f) What is the overall order of
the above reaction?
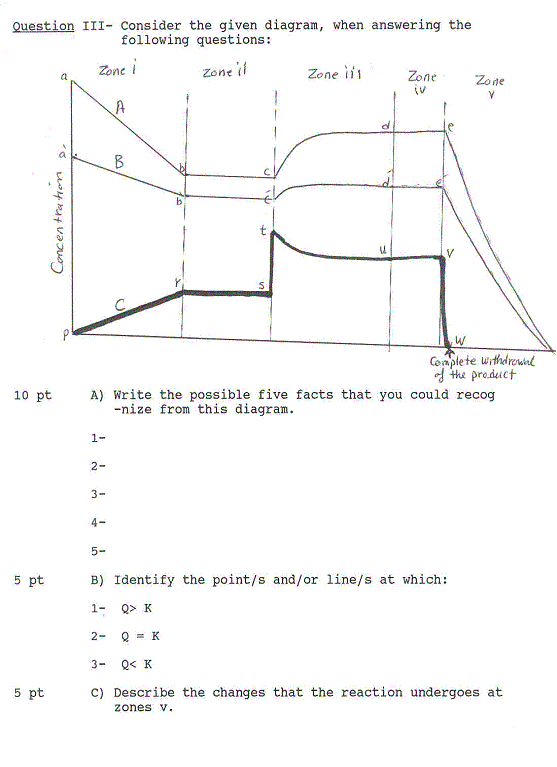
Question I11-Choose the correct answer(s):
1- For the reaction N2 + 3H2 ====== 2NH3 ,the relationship
between Kp and Kc
is given by:
a) Kp = Kcx(RT)-2 b)
Kc = Kpx(RT)-2 c) Kp =
Kc x (RT)2 d) Kc
= Kpx(RT) e) None
2- For the first order reaction which it's rate constant
is equal to 10-3 year-1 at 25 oC , the
half life time is,
a) 0.35 Yr. b)
693 year c) 0.457 year d) 8.316x103 month e) 12300 hour.
3- Which of the following is/are correct about the
catalytic reactions?
a) They are zero order b)
Homogeneous c) Exothermic
d) Nonspontaneous e)
Decreases the activation energy of the reactions
4- When Δ Go is negative, this means that:
a) K = 1
b) K > 1 c)
T = 0 d)
S = 0 e)
None
5- For the reaction 2AX (s) + BY(l) ------ A2B(l)
+ XB(g) .which of the following statement/s is/are correct:
a) This reaction is heterogeneous b)
Kc = PC
c) second order d) Unimolecular
e) Bimolecular
6- In part (5), Which of
the following is/are correct:
a) It is one step reaction mechanism b)Δ S(prod)> ΔS(react)
c) It is multi-step reaction mechanism d) Δ V is positive.
7- A
substances that formed in one
elementary step of a multi-step mechanism and consumed in another is known to be:
a) Reactant b) Product c) Intermediate d) Activated complex e) Transition state.
8- The
process which happen under constant pressure is known to be :
a) Isothermic b) Adiabatic c)
Isolated d) Isobaric e) Isochoric.
9- In the formula k = A e-Ea/RT symbol
A is known as :
a) Rate b) Order c) Frequency factor d) Activation energy e) Enthalpy.
10- The system which exchange both energy and matter with
the surroundings is known to be:
a)
Isothermal b) Spontaneous c) Open
d) Isolated e) Closed.

Question VI-Consider the given exothermic reaction when
answering the following question:
CH3CHO(l) +
Cl2(g) ====== CH3COCl(g) + HCl(g)
Obeys the following rate law, rate = k [Cl2]1/2[CH3CHO]
The following series of steps is proposed for this
reaction mechanism.
Step 1 Cl2 ========= 2 Cl.
Step 2 H.
+ Cl2
========= HCl
+ Cl.
Step
3 CH3CHO + Cl. ========= CH3COCl + H.
3 pt a) Write Kp
and Kc expression for the above reaction.
1 pt b) What is the overall reaction
order?
2 pt c) What is/are the intermediate/s in the mechanism?
2 pt d) Write the t0.5 - k relationship for this
reaction.
5 pt e) which of the steps above is the
rate determining step?
3 pt f)
What is the sign of ΔH,
ΔS, ΔG
2 pt g) Label fast steps.
2 pt h)
If the rate of this reaction decreases when
temperature increases, propose an explanation
for such behavior.
Question VII-
Answer as required:
10 pt A)
List five important facts that work as a bases of qualitative analysis. Give example that illustrates each fact:
(10 pts)
10 pt B)
Sketch a separation scheme for the mixture of Cu2+, Fe3+,
Al3+ ,Ni2+ and Mg2+,
Using the following reagents: HCl, H2S,
NH4Cl, NH4OH and NaOH.
(10 pts)
Question VIII- Explain
why: ( 4 pt
each )
1- Formation of two salts of H2CO3
such as NaHCO3, and Na2CO3, while H2SO4
doesn’t
2- NH4+ is stronger acid than Na+.
3- The mixture of equi 0.2M solution of sodium acetate
and acetic acid has higher buffer
capacity than the mixture of equi 0.5M NaCl
and HCl.
4- The pH of aqueous KCl solution is 7.
5-Addition of NH4 Cl(Cl-,NH4+)
to a solution of NH4OH (NH4+, OH-) will suppress its ionization.
Question IX:
10 pts A) Given that Ksp
of Pb(OH)2 is 1.2X10-15, and
the following dissociation :
Pb(OH)2 W Pb2+ +
2OH-
Calculate :
1- The concentration of Pb ions
in pure water.
2- The pH value of the suspension of Pb(OH)2 in water.
3- The
concentration of Pb ions in a solution of pH= 10.
10 pts B) Identify the fact/s that can be used to explain the
following separation:
1- Fe(OH)3/Pb(OH)2
+ excess NaOH
====> Pb(OH)3+ (aq) + Bi(OH)3
(PPT)
2- Cr3+ / Ni2+ + excess
NH4OH ===> Ni[NH3]42+
+ Cr(OH)3(ppt).
3- Ni2+/ Cu2+
+ ( HCl + H2S) ====> CuS(ppt) + Ni2+
4- Al3+/Ba2+ + (NH4Cl/NH4OH)
====> Al(OH)3(ppt)
+ Ba2+
Question X-
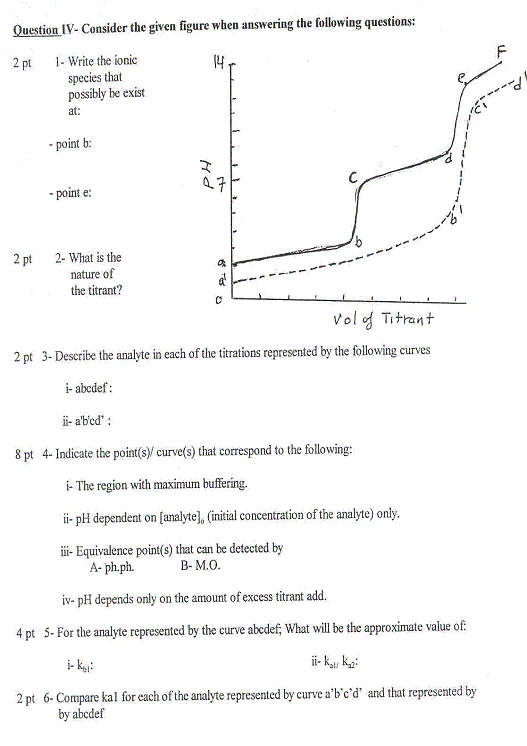
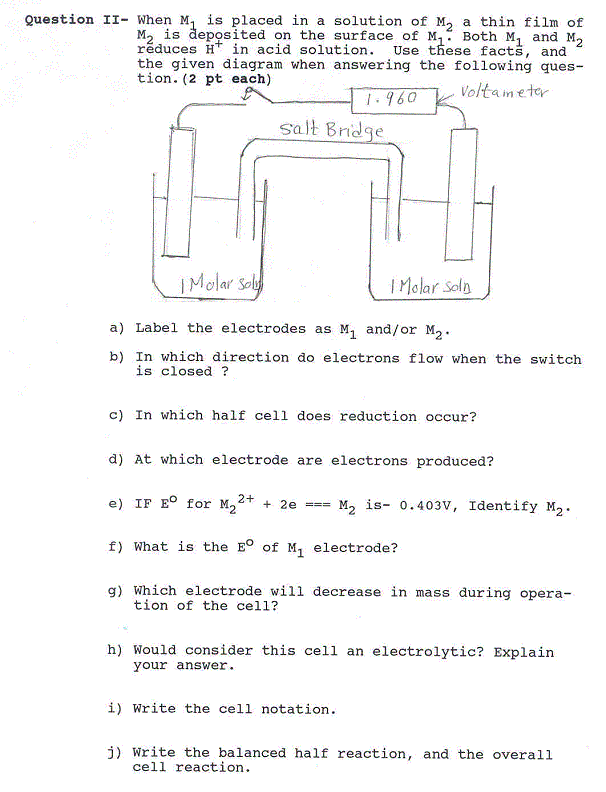
Question XI - Consider the given exothermic reaction and the accompanying
data when
answering the following
questions:
CO(g) + Cl2(g) =========
COCl2(g) Kc
= 4.57x109 at 100 oC.
2 pt a) Write the Kc
expression for this reaction.
2 pt b)
What is the value of Kp
at 100 oC.?
4 pt
c) Use Le Chatelier's principle to predict the
effect of the following on the position of the above reaction equilibrium.
1- Removal of some Cl2(g).
2-
Removal of some COCl2(g).
3-
Cooling the reaction container and it's contents.
4-
Decreasing the pressure.
2 pt d) Calculate >Go for this reaction at 100oC.
2 pt e) For the above reaction, if >Go 200oC is -109.48 KJ, what
will be the value of Kp .
6 pt f) Suppose the following
mechanism is correct for the above reaction.
step
1 Cl2 ==== 2 Cl. fast
step 2 Cl. + CO =====
COCl
fast
step 3 COCl + Cl2 =====
COCl2 + Cl. slow
step
4 2Cl. ===== Cl2 fast
Write the reaction rate (law) equation.
2 pt g) What is the overall order of
the above reaction?
Question XII- Solve the
following problems, ( 5 pt each )
1-
Balance the following equation, and identify the pH range required for this reaction.
I2 + NO3- = NO2(g) +
IO3-
2- If the
half life of 214Pb is 26.77 minutes, calculate the disintegration rate for 214Pb
.
3- Use the standard electrode potentials to calculate the ΔGo
for the following reaction,
Al(s) +
Fe2+(aq) ------< Al3+
+ Fe(s)
is this reaction spontaneous?
4-
Calculate the nuclear binding energy of 2713Al3+ nucleus, given that mass is 2713Al3+ 26.98 amu.
Question XIII-Choose the correct answer(s):
1- For the reaction N2 + 3H2 ======
2NH3 ,the relationship between Kp
and Kc is given
by:
a) Kp = Kcx(RT)-2 b) Kc =
Kpx(RT)-2 c) Kp =
Kc x (RT)2 d) Kc
= Kpx(RT) e) None
2- For the first order reaction which it's rate constant
is equal to 10-3 year-1 at 25 oC , the
half life time is,
a) 0.35 Yr. b) 693 year c) 0.457 year d)
8.316x103 month e) 12300
hour.
3- Which of the following is/are correct about the
catalytic reactions?
a) They are zero order b)
Homogeneous c) Exothermic
d) Nonspontaneous e)
Decreases the activation energy of the reactions
4- When Δ Go is negative, this means that:
a) K = 1
b) K > 1 c)
T = 0 d)
S = 0 e)
None
5- For the reaction 2AX (s) + BY(l) ------ A2B(l)
+ XB(g) .which of the following statement/s is/are correct:
a) This reaction is heterogeneous b)
Kc = PC
c) second order d) Unimolecular
e) Bimolecular
6- In part (5), which of the following is/are
correct?
a) It is one step reaction mechanism b)Δ S(prod)> ΔS(react)
c) It is multi-step reaction mechanism d) Δ V is positive.
7- Substances that formed in one elementary step of a
multi-step mechanism and consumed in
another is known to be:
a) Reactant b) Product c) Intermediate d) Activated complex e) Transition state.
8- The
process which happen under constant pressure is known
to be:
a) Isothermic b) Adiabatic c)
Isolated d) Isobaric e) Isochoric.
9- In the formula k = A e-Ea/RT symbol
A is known as :
a) Rate b) Order c) Frequency factor d) Activation energy e) Enthalpy.
10- The system which exchange both energy and matter with
the surroundings is known to be:
a)
Isothermal b) Spontaneous c) Open
d) Isolated e) Closed.
Question XIV: Explain why:
1- In the following cell
Ag/Ag+
2 Cd/ Cd 2+ Ag
electrode may lose weight.
2- The nuclear reaction :
P( proton) ===> n (neutron ) + +e
(Positron) Is endoergic ,While
P( proton) + e (K-shell)
====> n Is exoergic.
3- K metal cannot be generated from the aqueous
electrolytes.
4- Hydrolysis of aqueous HBr, produces H2
gas at the cathode and Br2 gas at the anode,
while that of aqueous HF produces H
2 gas at the cathode, and O2 gas at the anode.

Question XV- Write the major difference/s between: (4 pt each)
1- Natural radioactivity and nuclear transmutation.
2- Galvanic (Voltaic) cell Electrolytic cell
3- Mechanical (paints) and cathodic protection from
corrosion.
4- Positron emission and K- electron capture.
5- Electrolysis of aqueous solution of NaCl and aqueous CuCl2.
Question XVI - Answer each of the following with true or false.
If a statement is false correct it.
----- 1- Eo for the cell CdCd2+ Ni2+ Ni, is positive.
----- 2-
For conversion of proton to neutron , it is easier
through k electron capture
rather than positron emission.
----- 3- A car battery during charging
process is an example of Galvanic (Voltaic) cell.
----- 4- A mass change of 1gram in nuclear reaction is
equivalent to 934.31 Mev.
----- 5-
Nuclei of odd numbers of protons and even numbers neutrons are generally more stable than those which have even numbers of
protons and even numbers
of neutrons.
----- 6-The following is a notation for Electrolytic cell:
AgAg+Zn2+Zn.
----- 7-
Both Al and Cu metals can be produced by electrolysis of their aqueous solutions.
----- 8-
The amount of radiation that deposits 1x10-2 J of energy per Kg of
tissue, is the definition of rem.
----- 9- Nucleus that has more than 230 nucleons adjust
n/p ratio by emitting β particles.
----- 10- The
radioactive decay of 23892 U to Pb
is considered to be second order reaction.
Question XVII- Choose the correct answer/s: ( 2 pt each )
1- Each of the following is weaker than HCl except:
a) H2CO3 b)
HClO2 c) HBr. d)
HClO3 e)HI
2- Each of the following has a common ion with NaHSO3
except
a)NaHSO4 b) Na2SO3 c) H2SO3 d) CaS2O3 e) H2SO4.
3- Aqueous solutions of NH4Cl is:
a)
Acidic b)
Alkaline c) Neutral d) Basic e)
None
4- For alcoholic solution of 10-3
M HCl, the pOH is:
a) 12 b)
10.56 c) 11 d) -log Ka e)
none.
5- Which of the following is a conjugate base of HPO4
2- ?
a) PO43- b) H3PO4 c)
H2PO4- d) H3PO3 e)H2PO3-
6-
" The amount of acid/ base a buffer can neutralize before the pH begins to
change appreciably" is
the definition of:
a)
Strong acid b) Amphoteric
substances c) Lewis base
d) Buffer solutions e)
Buffer capacity
7- Acid strength is determined by:
a) Acid decomposition b) Acid
stability c) ka d) #of H+/molecule e) Acid origin.
8- Precipitation occurs when:
a) Ksp= 10-14 b) Ksp
< Q (ionic product) c) Q
< Ksp d) Ksp
= Q e) Q = 10-14
9-
Metal hydroxides/oxides which dissolve in strongly acidic or strongly basic
media are known to be:
a) Salts b)
Amphoteric
c) Strong acids d)
Weak bases e) polyprotic acids
10- Which of the following is a Lewis
base?
a)
HCl b) BCl3 c)
CO32- d) NH3 e)
H3O+
Question XVIII- What is/are the difference/s
between: (5 pt each )
1- Electrochemical series and Spectrochemical series.
2- High spin complex and low spin complex.
3- H2NCH2CH2NH2
and CN- as a ligand.
4- Alkene and Alkyne.
5- Tetrahedral and Octahedral complexes.
Question XIX- A)
Write the structural formula for each of the following: (3 pt each)
a) 2,2 Dimethyl-,6,6 Dibromo 3- Heptyne.
b) cis
2,3 Dichloro 2-Pentene.
c) Hexammine
Chromium(III) Nitrate.
d) Sodium Tetrabromo (ethylenediamine) Cobaltate(III).
e) 2-Amino 3,4 di-methyl Hex -3-ene.
B- Choose the correct answer(s) :(10 pt )
1- In each of the following, the value of Δ is higher than spin pairing
energy except:
a) Fe(CN)63- b) NiCl42- c) Cr(CN)63- d) CoF63- e) Mn(CN)6
4-.
2- The oxidation number of Chromium in [Cr(NH3)4Cl2]Cl is:
a) -3 b) 6
c) 2 d) -2 e) None
3- Which of the following complexes has coordination number of 6.
a) [Zn(NH3)2Cl2] b) K3[Fe(CN)6] c) [Cu(NH3)4]2+
d) [Co(NH3)5Cl]2+
e)Pt(NH3)2Cl2.
4- Complexes which have higher number of unpaired
electrons are known to be:
a) Inert b) paramagnetic c) diamagnetic d) low spin e)
high spin.
5- The functional group/s in NO2-CH2=CH-CH2
-CO-CH2CH2OH is/ are:
a) Nitrile, Ketone, Ether and Aldehyde.
b) Nitro , Alkene, Ether and Aldehyde.
c) Nitro, Alkene
, Ketone and Alcohol.
d) Alcohol, Alkene ,
Ether and Nitro.
e) Amino , Alcohol, Alkyne and Ketone.
Question XX- A) Write the major difference/s between:(
2.5 pt each)
1- Cathodes in galvanic or voltaic cell and Cathodes in electrolytic cell.
2- Positron emission and K- electron capture.
B) Choose the correct answer/s: ( 2
pt each )
1- Each of the following is weaker than HCl except :
a) H2CO3 b) HClO2 c) Hbr. d) HClO3 e)HI
2- Each of the following has a common ion with NaHSO3
except
a)NaHSO4 b) Na2SO3
c) H2SO3 d) CaS2O3 e) H2SO4.
3- Aqueous solutions of NH4Cl is:
a) Acidic b)
Alkaline c) Neutral d) Basic
e) None
4-
For alcoholic solution of 10-3 M HCl, the pOH is:
a) 12 b) 10.56 c)
11 d)
-log Ka e)
none.
5- Which of the following is a conjugate base of HPO4 2-
?
a) PO43- b) H3PO4
c) H2PO4- d)
H3PO3 e)H2PO3-
6- " The amount of acid/ base a buffer can
neutralize before the pH begins to change appreciably"
is the definition of:
a) Strong
acid b)
Amphoteric substances
c) Lewis base
d) Buffer
solutions e) Buffer capacity
7- Acid strength is determined by:
a) Acid decomposition b) Acid stability c) ka
d) #of H+/molecule e) Acid
origin.
8- Precipitation occurs when:
a) Ksp= 10-14 b) Ksp < Q (ionic product)
c) Q < Ksp d) Ksp = Q
e) Q = 10-14
9-
Metal hydroxides/oxides which dissolve in strongly acidic or strongly basic media
are known to be:
a) Salts b) Amphoteric c) Strong acids d) Weak bases e) polyprotic
acids
10- Which of the following is a Lewis
base?
a) HCl b)
BCl3 c)
CO32- d)
NH3 e)
H3O+




